 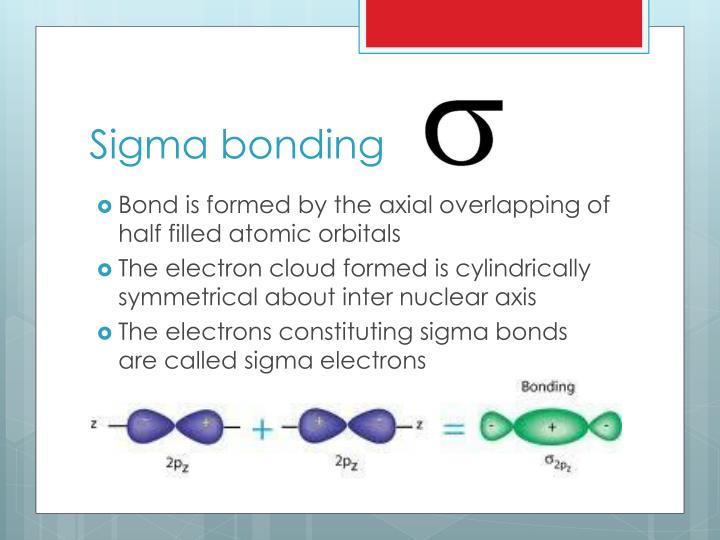
As a result, double and triple bonds with pi bonds exhibit geometric (cis-trans) isomerism. Restricted Rotation: Pi bonds restrict the rotation of atoms around the bond axis because breaking the pi bond requires the electrons to be reconfigured.For example, in H2C=CH2 (ethylene), the bond between the two carbon atoms is a sigma bond, while the two bonds between the carbon and hydrogen atoms are pi bonds. The common forms of sigma bonds are shown below. A bond comes from the end-to-end overlap of the bonding orbitals. A bond has cylindrical symmetry a bond has a nodal plane that includes the bond axis. Double and Triple Bonds: In molecules with double or triple bonds, one or more of the bonds typically involve pi bonds. What is the difference between a sigma bond and a pi bond Medium.Weaker and Less Stable: Pi bonds are generally weaker and less stable than sigma bonds due to the less effective overlap of orbitals.The electron density is concentrated above and below the internuclear axis. The electrons drawn to the nuclei of both atoms, and the force of attraction between the two nuclei and the shared electrons holds the atoms together. 
sigma and pi bonds and correctly predicts the geometry of molecules 24 CLEP CHEMISTRY. A sigma bond is a type of covalent bond that is formed when two atoms share a pair of electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
